and Canada) or 72 (outside US and Canada).
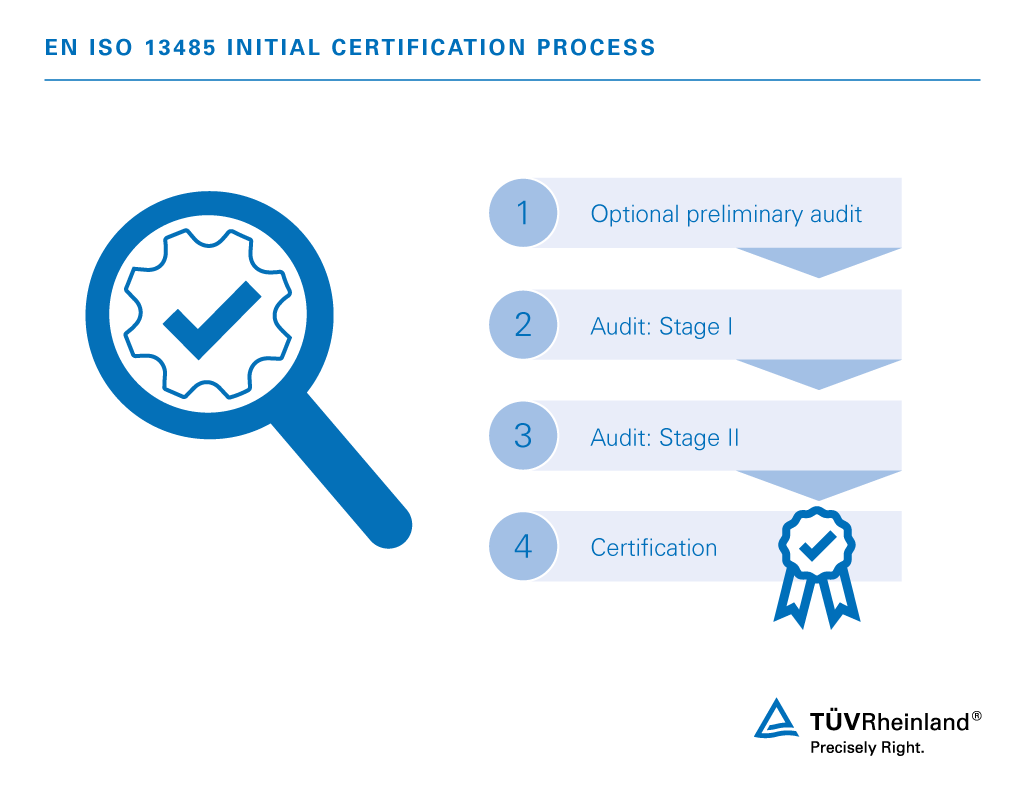
Iso 13485 standards iso#
Iso 13485 standards upgrade#
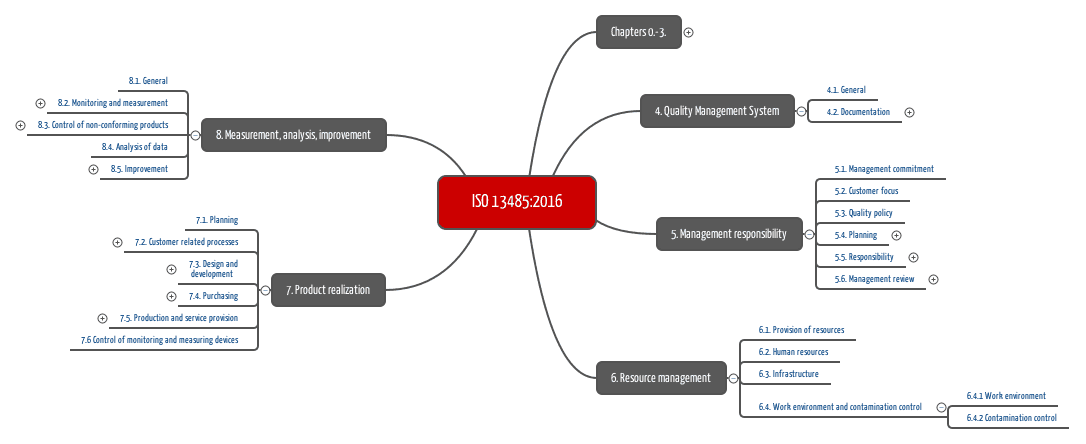

Safety check # 2: CE labelĮveryone has heard about it, but most do not know what it means: the CE label, without which no product whatsoever can be placed on the market in the European Union. This discerning quality management system has a decisive advantage for our clients: they can rely on the entire product range to correspond with the highest quality and safety standards. Compared to the latter however, it sets out additional requirements such as, among other things, product traceability, introduction of risk management processes, the validation of manufacturing software, maintenance and monitoring, validation of product design and development as well as measures for sterility and cleaning. ISO 9001 is the international standard which provides specifications for a quality management system which can be applied at any organization regardless of industry, product or service, or company size. In many aspects, the DIN EN ISO 13485:2012, as it is called in its current version, is congruent with the international standard for quality management, the DIN EN ISO 9001. The ISO 13485 standard is widely accepted as the benchmark for medical device manufacturers quality management systems. The primary difference between ISO 13485 and ISO 9001 for medical devices is the scope of these quality standards.
